- Menu
- Search For Product Models
Search For Product Models
- cn
Selecting the proper material for a heat exchanger is critical to ensure long-term reliability, corrosion resistance, and operational safety.
Material choice depends primarily on:
Operating temperature
pH value of the medium
Chloride (Cl⁻) concentration (ppm)
Pressure conditions
Presence of oxidizing agents
This article compares five commonly used materials in industrial heat exchangers: 304, 316L, Titanium (Ti), 254 SMO, and Hastelloy C276.
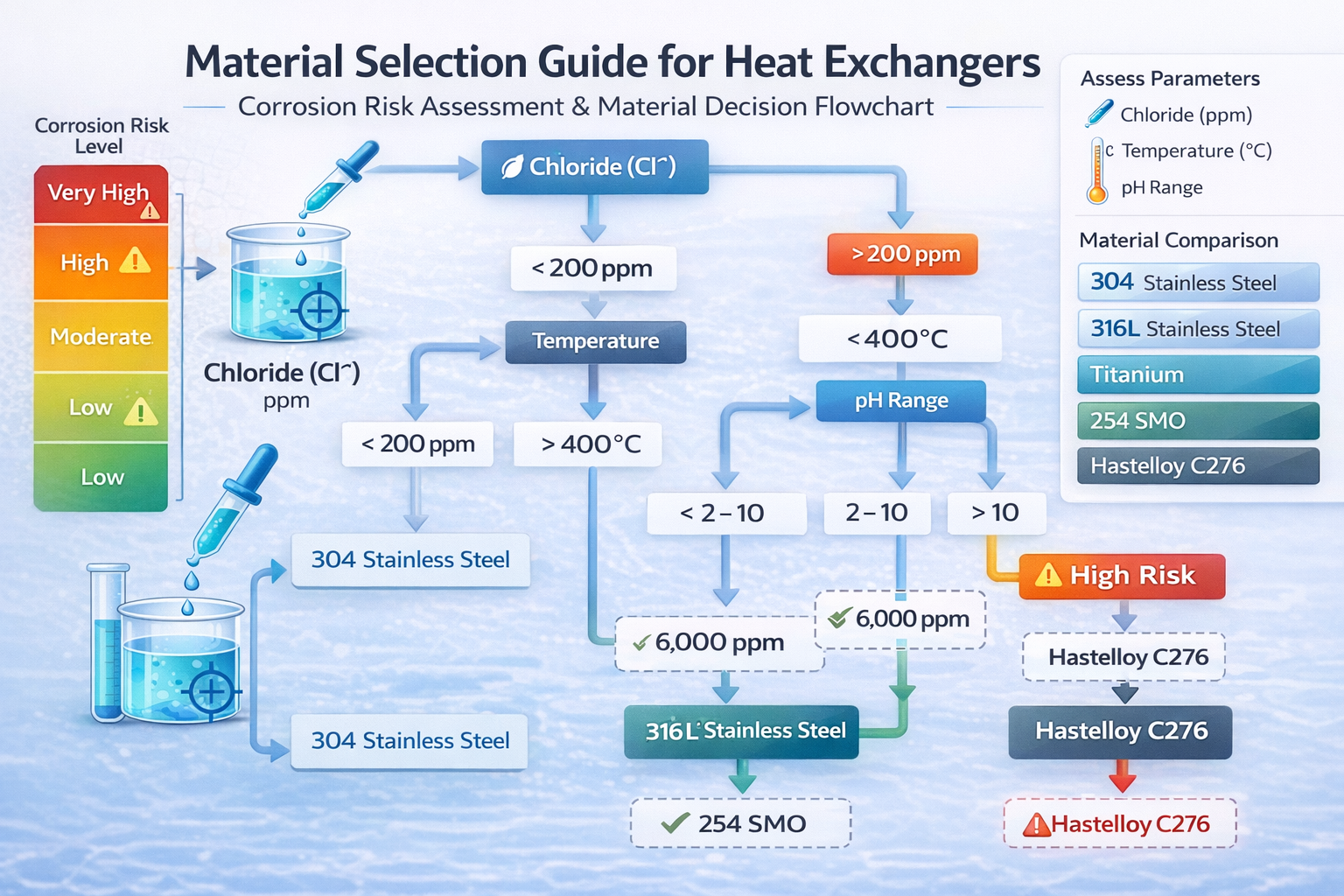
One table view for temperature, pH range, chloride limit, and typical heat exchanger applications.
| Material | Temperature Range | pH Range | Chloride (Cl⁻) Resistance | Typical Applications | Notes / Warnings |
|---|---|---|---|---|---|
| 304 Economical | Recommended: up to 400°C (dry) In chloride: limited to < 60°C | Neutral: pH 6 – 9 Not recommended for strong acids/alkali | Safe limit: < 200 ppm Prone to pitting / SCC in chloride | Fresh water systems / Food processing / HVAC (controlled water quality) | ⚠ Not suitable for seawater, saltwater pools, aggressive chemical media |
| 316L General purpose | Recommended: up to 400°C In chloride: typically < 80°C | pH 2 – 10 (depends on chloride & temperature) | Safe limit: < 1000 ppm (low temp) Risk of pitting above 60–80°C | Mild seawater exposure / Pharmaceutical processing / Pool HX (freshwater only) | ⚠ Not ideal for high-salinity or high-temperature chloride environments |
| Titanium (Gr1/Gr2) Seawater | Recommended: up to 250°C Excellent stability in wet chloride | Effective in pH 1 – 12 Excellent resistance to oxidizing acids | Practically immune to pitting Suitable for seawater (>20,000 ppm) | Seawater heat exchangers / Saltwater pools / Desalination / Marine | ✔ Preferred material for saltwater pool heat exchangers |
| 254 SMO High Cl⁻ | Recommended: up to 400°C Chloride: safe up to 100°C (depends on ppm) | pH 2 – 11 | Resistant up to ~6,000 ppm (low temp) Much higher pitting resistance than 316L | Brackish water / Offshore platforms / Chemical processing / Medium salinity | ✔ Alternative to titanium in moderate seawater conditions |
| Hastelloy C276 Extreme chemistry | Up to 1000°C (dry) Highly stable in high-temp corrosive env. | pH 0 – 14 Resistant to strong acids (HCl, H2SO4, etc.) | Excellent chloride-induced corrosion resistance Suitable for high chloride + high temperature | Acid processing / Chemical reactors / Highly corrosive industrial systems | ✔ Premium material for extreme conditions |
| Material | pH Range | Max Cl⁻ (ppm) | Temperature Limit | Typical Use |
|---|---|---|---|---|
| 304 | 6–9 | <200 | <60°C (chloride) | Fresh water |
| 316L | 2–10 | <1000 | <80°C (chloride) | Mild corrosive |
| Titanium | 1–12 | Seawater safe | <250°C | Marine / Pools |
| 254 SMO | 2–11 | ~6000 | <100°C (chloride) | Brackish water |
| C276 | 0–14 | Very high | <400°C+ | Chemical industry |
Material selection should consider:
Chloride concentration first
Operating temperature second
pH range third
Budget and lifecycle cost
For swimming pool applications:
Freshwater → 316L
Saltwater → Titanium
Brackish water → 254 SMO
For chemical plants:
Acidic media → C276
Material selection for heat exchangers is a balance between corrosion resistance, mechanical strength, temperature tolerance, and cost.
While 304 and 316L are economical solutions for controlled environments, titanium and high-alloy materials like 254 SMO and C276 provide superior durability in aggressive conditions.
Proper analysis of pH, chloride concentration, and temperature is essential to prevent premature corrosion and extend service life.
304 stainless steel is generally safe below 200 ppm chloride concentration in low-temperature conditions.
Above this level, the risk of pitting corrosion increases significantly.
316L may start experiencing pitting corrosion above 1,000 ppm chloride, especially when temperatures exceed 60–80°C.
Higher temperatures accelerate corrosion risk.
Titanium is highly resistant to chloride-induced pitting and stress corrosion cracking.
It is suitable for seawater applications (>20,000 ppm Cl⁻), but scaling can still occur if water chemistry is not controlled.
254 SMO is recommended for moderate to high chloride environments (up to approximately 6,000 ppm at lower temperatures).
It offers significantly better pitting resistance than 316L.
Hastelloy C276 is selected for extreme environments:
Strong acids
High chloride + high temperature
Chemical process industry
It is usually unnecessary for standard water or pool heating systems due to high cost.
Yes.
In neutral water systems, chloride concentration is the main driver of corrosion.
In strong acidic environments (low pH), corrosion risk increases even if chloride levels are moderate.
Corrosion rates increase exponentially with temperature.
A material that performs safely at 40°C may fail at 90°C under the same chloride concentration.
304 is generally not recommended for chlorinated or saltwater pools.
Even moderate chloride levels can cause premature failure.
254 SMO can replace titanium in some brackish water applications.
However, for full seawater exposure or highly aggressive chloride conditions, titanium remains more reliable.
For water-based systems:
Chloride concentration
Operating temperature
pH range
Chloride + temperature together determine pitting resistance requirements.
Consider:
Maximum (not average) chloride concentration
Peak operating temperature
Oxygen content
Flow velocity
Design life
Conservative material selection reduces lifecycle cost.
At elevated temperatures, chloride ions penetrate the passive film more easily, leading to pitting and stress corrosion cracking.
Freshwater pool → 316L
Saltwater pool → Titanium
Brackish water → 254 SMO
Chemical process → C276